Bohr Diagram For Beryllium General Wiring Diagram
enhance student understanding of atomic structure. A Bohr model, more correctly a deBroglie-Bohr model, is used here to calculate the total electronic energies of atoms and ions containing up to four electrons. The Bohr model for the hydrogen atom is the prototype of the semi-classical approach to atomic and molecular structure.
Labelled Diagram Of An Atom Of Beryllium Labeled Diagram
6.2 The Bohr Model; 6.3 Development of Quantum Theory; 6.4 Electronic Structure of Atoms (Electron Configurations) 6.5 Periodic Variations in Element Properties;. the beryllium and boron atoms each have only four and six electrons, respectively. It is possible to draw a structure with a double bond between a boron atom and a fluorine atom in.

Atomic Structure (Bohr Model) for Beryllium (Be) YouTube
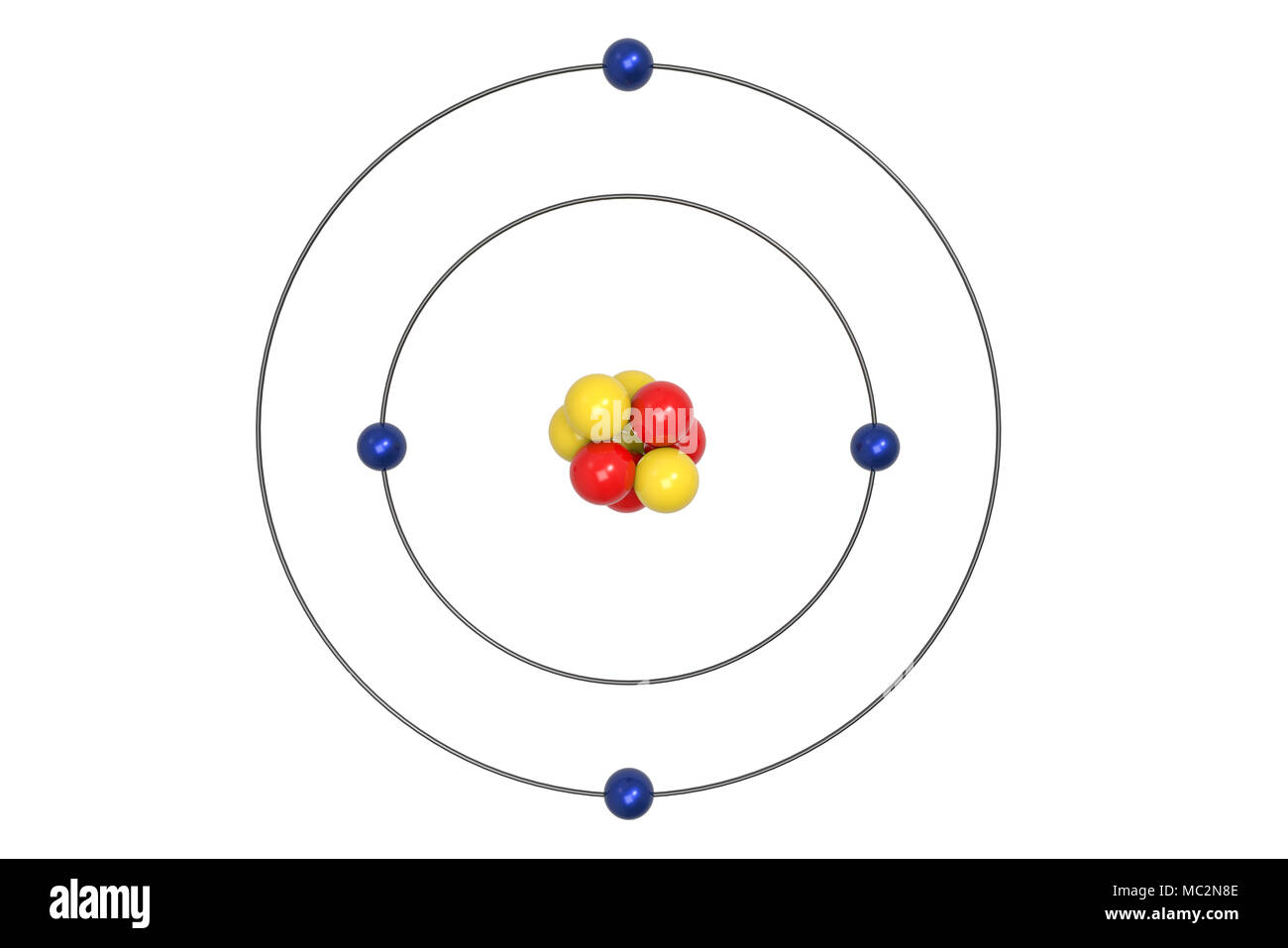
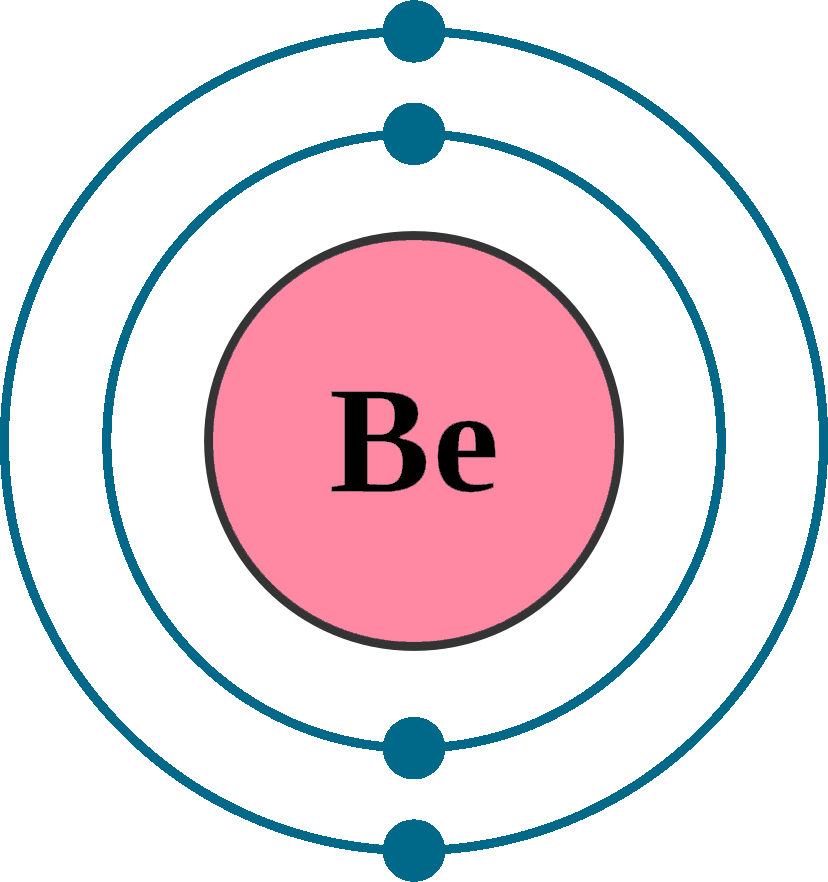
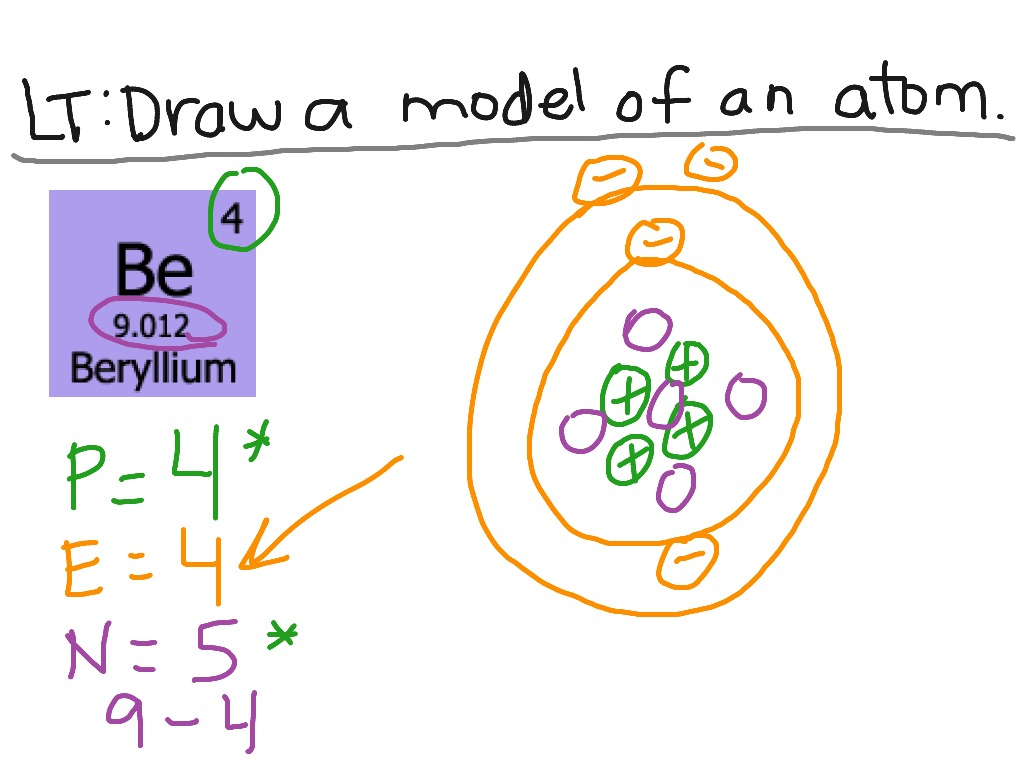
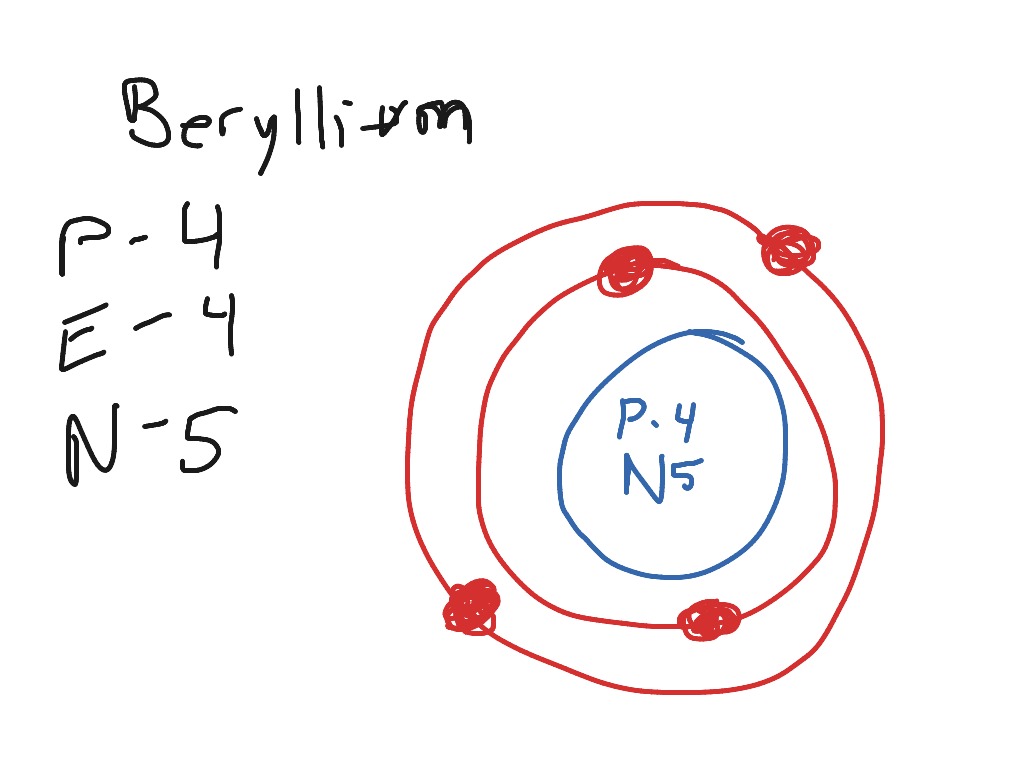
Steps Write protons, neutrons, and electrons of beryllium atom Beryllium has 4 protons, 5 neutrons, and 4 electrons. Learn how to find: Beryllium protons neutrons electrons Draw nucleus of beryllium atom The nucleus of a beryllium atom contains 4 protons and 5 neutrons. So draw the nucleus of beryllium atom as follows: Beryllium nucleus
Beryllium Bohr Model Diagram
Bohr model of sodium atom; beryllium atom Bohr model; In 1913, Danish physicist Neil Bohr proposed the Bohr atomic model based on Planck's quantum theory of radiation. This atomic model is the modification of Rutherford's atomic model (the nucleus is positively charged and is surrounded by electrons (negatively charged particles).
Beryllium Bohr Model Diagram
34 4.8K views 1 year ago In this video we'll look at the atomic structure and Bohr model for the Beryllium atom (Be). We'll use a Bohr diagram to visually represent where the electrons are.
Beryllium Element With Reaction, Properties and Uses Periodic Table
The Bohr model explains the stability of the atom and atomic particles. It also talks about the position of various atomic particles inside the atom as well as their charge and other properties. It describes the structure of the atom in detail.

Beryllium Bohr Diagram
The calculation for the beryllium atom is carried out as shown below. Required student input is indicated by the highlighted regions. Enter nuclear charge: Z := 4 Kinetic energy: T1 ( R1 ) 1 := 2 ⋅ R12 1 T2 ( R1 ) := 8 ⋅ R12 Electron‐nucleus potential energy: − Z VN1 ( R1 ) := R1 − Z VN2 ( R1 ) := 4 ⋅ R1
Beryllium Bohr Diagram exatin.info
The Bohr model is a relatively primitive model of the hydrogen atom, compared to the valence shell model. As a theory, it can be derived as a first-order approximation of the hydrogen atom using the broader and much more accurate quantum mechanics and thus may be considered to be an obsolete scientific theory.
Beryllium model Stock Vector Images Alamy
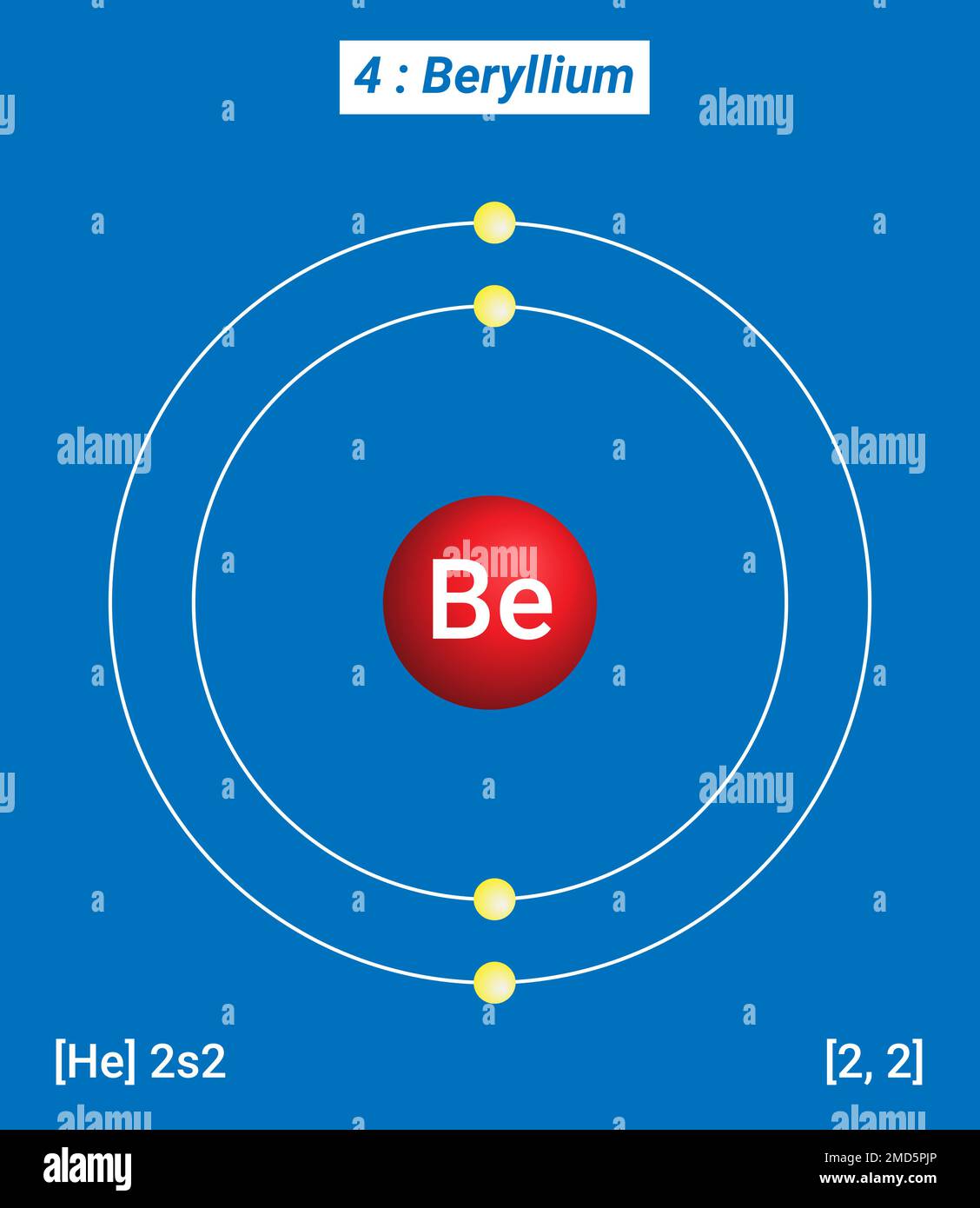
The Bohr Model for Beryllium (Be) has 4 protons in the nucleus due to the atomic number of Be being 4. The Mass number is 9 which means Beryllium needs 5 neutrons in the nucleus. (Mass number = protons + neutrons, 9 = 4 + n). Beryllium has four electrons to balance the four protons.

Bohr Diagram For Beryllium General Wiring Diagram
Electrons arrangement or Bohr model: 2, 2: Electronic configuration [He] 2s 2: Atomic radius: 153 picometers (van der Waals radius) Valence electrons: 2: 1st Ionization energy: 9.323 eV:. You have already seen the Bohr model of Beryllium atom in the above table and you have seen that the number of orbits or shells in sodium is 2.
Modelo Bohr de Átomo de Berilio con protones, neutrones y electrones
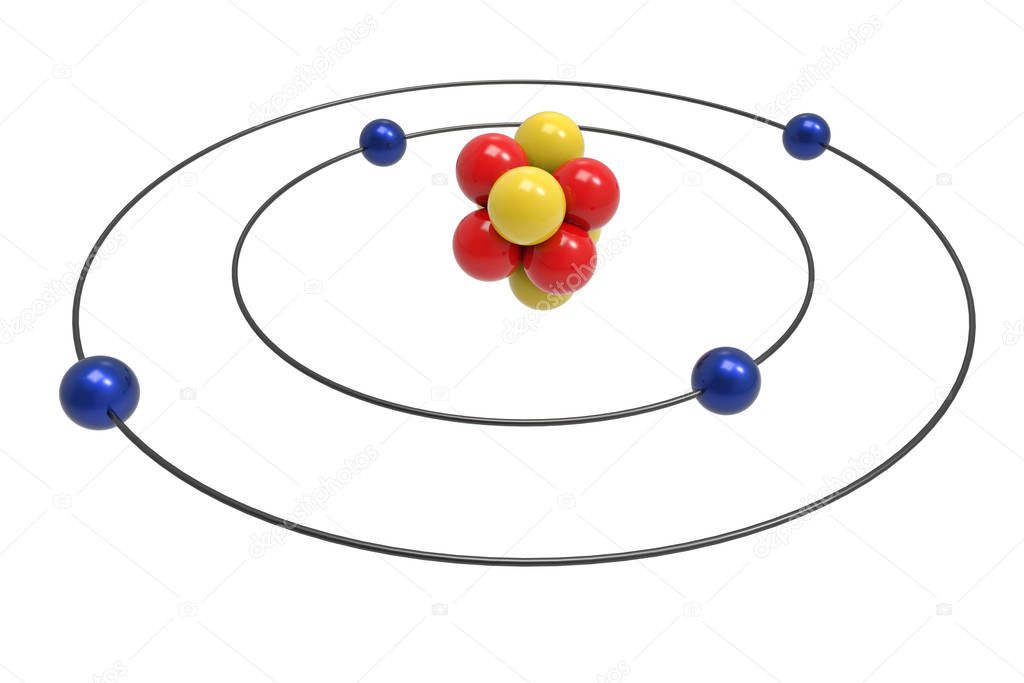
The model shown in the figure below is for the beryllium atom or any four‐electron ion. Note that the occupancy of the inner orbit is restricted to two electrons (Pauli Principle) and that the orbit radii are constrained by the hydrogen atom result (R 2 = 4R 1 ) leaving only one variational parameter, the radius of the n = 1 orbit.

Beryllium Bohr Diagram Transborder Media
Atomic & Molecular Structure How to Make a 3-D Bohr Model ••• Updated April 24, 2017 By Tricia Lobo In your introductory chemistry classes you will have to become familiar with a number of the early models of atoms, which represent scientists' early concepts of the structure of atoms.

Beryllium Bohr Model
Ai = hν = hc λ A i = h ν = h c λ. Unknown wavelength λ λ can be determined from the Rydberg formula: 1 λ =R∞Z2( 1 n21 − 1 n22) 1 λ = R ∞ Z 2 ( 1 n 1 2 − 1 n 2 2) so that final equation for ionization energy looks like this: Ei = hc e R∞Z2( 1 n21 − 1 n22) E i = h c e R ∞ Z 2 ( 1 n 1 2 − 1 n 2 2)
Beryllium Bohr Diagram exatin.info
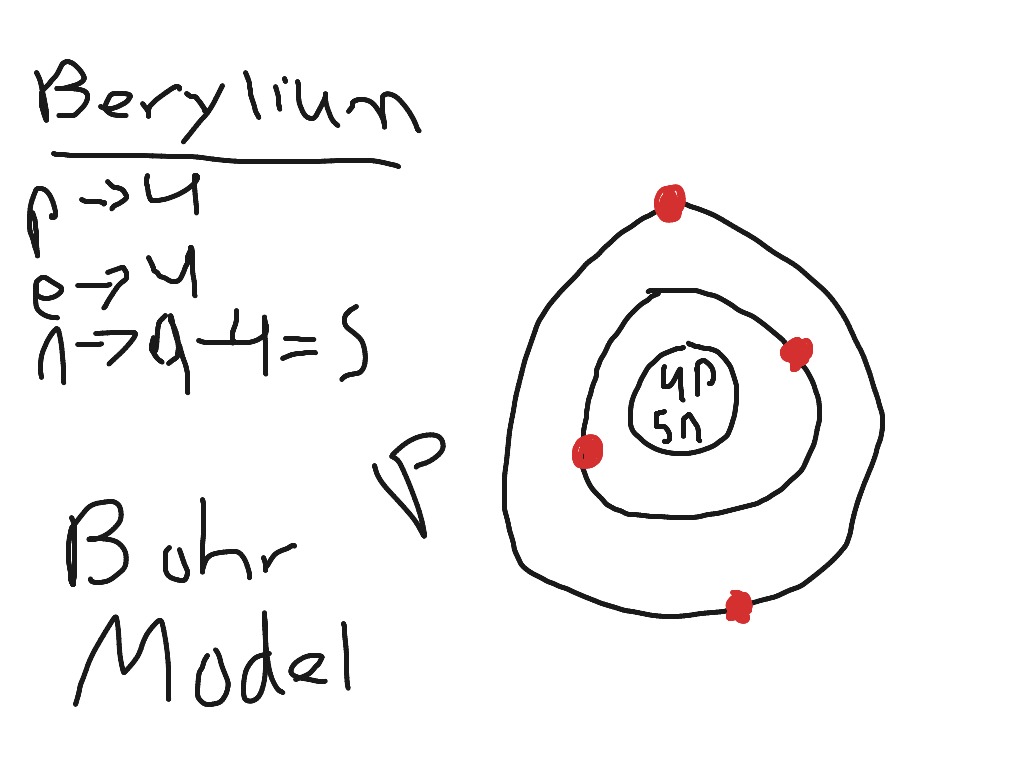
The Bohr Model of Beryllium (Be) has a nucleus that contains 5 neutrons and 4 protons. This nucleus is surrounded by two-electron shells named K-shell and L-shell. The outermost shell in the Bohr diagram of Beryllium contains 2 electrons that also called valence electrons. Page Contents show How to draw Bohr Model of Beryllium (Be)?
Beryllium Bohr Diagram
Bohr model of Elements. 1. Hydrogen (H) 1. 2. Helium (He) 2. 3. Lithium (Li)
ShowMe beryllium bohr model
Build an Atom - PhET Interactive Simulations